
Research

Research background
Optical imaging has played an essential role in studying various physical, chemical and biological systems by visualizing their structures, dynamics, and functionalities. The research trend for the previous decade is aimed at improving optical resolving power, and significant advances have been made, resulting in an improved spatial resolution that is more than an order of magnitude better than the diffraction limit. This progress has made a tremendous impact on the in vitro studies of single molecules and individual biological cells. However, the use of optical imaging for in vivo applications of interrogating living tissues, which holds promise in the life sciences such as neuroscience and early medical diagnosis, is still in needs of improvements. At the current stage of development, the fundamental limitation is set by the shallow working depth of optical imaging in biological tissues due to the multiple elastic light scattering induced by the tissues.
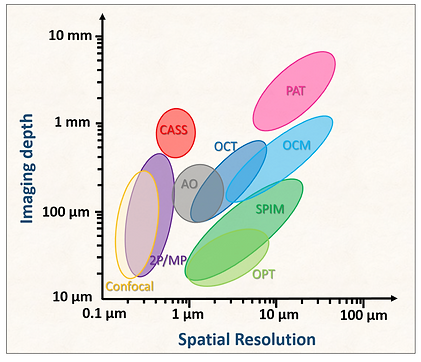
There have been numerous studies that have attempted to resolve the detrimental effect of multiple light scattering. However, their performance in spatial resolution is compromised with an increase of target depth to such an extent that the ratio of imaging depth to spatial resolution stays around 100 (Figure below). Those approaches that can reach deep inside tend to lose spatial resolution because they rely on the statistical analysis of multiple light scattering, rather than the microscopic details of the scattering events. The approaches covering the other extreme of high spatial resolution tend to be near-sighted because of their inability to tell single- from multiple-scattered waves. With the increase of target depth, the signal strength of single-scattered waves decays exponentially at a length scale set by the scattering mean free path, which is on the order of 100 microns in biological tissues. Conventional high-resolution optical imaging modalities were, therefore, unable to cope with the decreasing ratio of single scattering signal to the multiple scattering backgrounds. First-order corrections have been made to the low-order Zernike polynomial in adaptive optics, but the resulting improvement has been marginal.

Research objective
The main aim of our lab is to develop super-depth optical methodologies for imaging, sensing, and light manipulation of targets located deep within scattering media. We will develop optical imaging systems that can cover the dimensions of space, momentum, and time of both incidence and scattered waves. With this unprecedented level of detail on the response of the scattering media, we will solve inverse scattering problems at the microscopic level of scattering events, and identify the time-dependent modes of the scattering media. In doing so, we will achieve super-depth imaging at the microscopic resolution for the elastic and inelastic scattering, and deliver light energy that is two orders of magnitude, or more, larger than currently existing methods. At every step of the process, we will seek to apply the developed methods to important biological and biomedical applications.